|
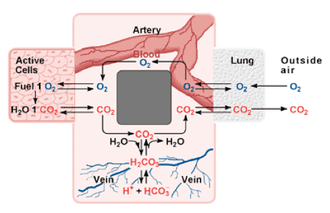
Buffers are substances that minimize changes in the concentrations of H+ and OH- in a solution. The carbonic acid buffer system in human blood is an example. Carbonic acid can give away H+ if there are too many OH- in the blood and can also take back H+ if there are too many H+ in the blood.
|
Helpful Online Resources: