|
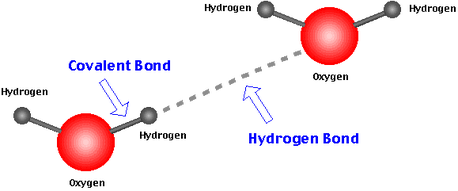
Hydrogen bonds are the bonds between water molecules and make water's special properties possible by ordering molecules into a higher level of structural organization. Hydrogen bonds are possible because water is a polar molecule, meaning that opposite ends of the molecule have opposite charges. The charged portions of a water molecule are attracted to oppositely charged parts of neighboring molecules. The hydrogen atom of one water molecule is slightly positive and is attracted to the slightly negative oxygen atom of a neighboring molecule. This allows molecules to hydrogen-bond to multiple molecules, and these associations are constantly changing.
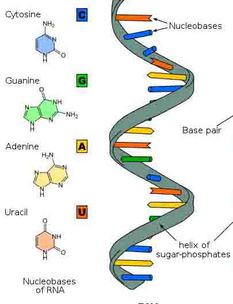
Hydrogen bonds are also important to molecules other than water. They are extremely important to RNA and allow RNA to perform many different functions. For example, RNA can hydrogen-bond to other nucleic acid molecules. In addition, RNA can assume a specific three-dimensional shape by forming hydrogen bonds between bases in different parts of its own polynucleotide chain. Hydrogen bonds are very important to RNA and are part of what make the molecule so versatile. |
Helpful Online Resources: