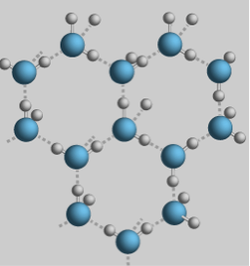
The expansion of water upon freezing is vital to life on Earth. It is what causes ice to be less dense as a solid than as a liquid. This means that ice floats in liquid water. Hydrogen bonding is once again the source of this rare property. Water freezes when its molecules are no longer moving around enough to break their hydrogen bonds. When water freezes it becomes locked into a crystalline lattice, and each water molecule bonds to four neighboring molecules. The hydrogen bonds allow for the molecules to be kept far enough apart so that ice is less dense than liquid water at 4 degrees Celsius.This may not seem like a very important property, but the fitness of the environment would drastically change if water was less dense than ice. All of bodies of water would eventually freeze over if ice did sink, essentially making life on Earth impossible.

Helpful Online Resources:
