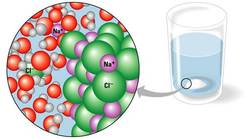
 Salt dissolving in water. A hydration shell surrounds each solute ion.
Salt dissolving in water. A hydration shell surrounds each solute ion.
A solution is a liquid that is a completely homogeneous mixture of multiple substances. (An aqueous solution is one in which water is the solvent.) The substance that is dissolved in a solution is called the solute. The solvent is the substance that dissolves the solute. Water is the best solvent. However, water cannot dissolve everything. Water's versatility as a solvent can be traced back to its polarity/hydrogen bonds. the hydrogen side of a water molecule has a positive charge while the oxygen side has a negative charge, which allows water molecules to become attracted to many different types of molecules. For example, in NaCl, water molecules surround the individual sodium and chloride ions to form hydration shells. Hydration shells are the spheres of water molecules around each dissolved ion. Finally, the water eventually dissolves all of the ions. Substances that are attracted to water are hydrophilic, while those that are nonionic and nonpolar are hydrophobic because they repel water. A colloid is a stable suspension of fine particles in a liquid. Colloids are created when there are hydrophilic substances within water that do not dissolve. Molarity is the number of moles of solute per liter of solution. It is the unit most often used to measure concentration by scientists.
Helpful Online Resources:
